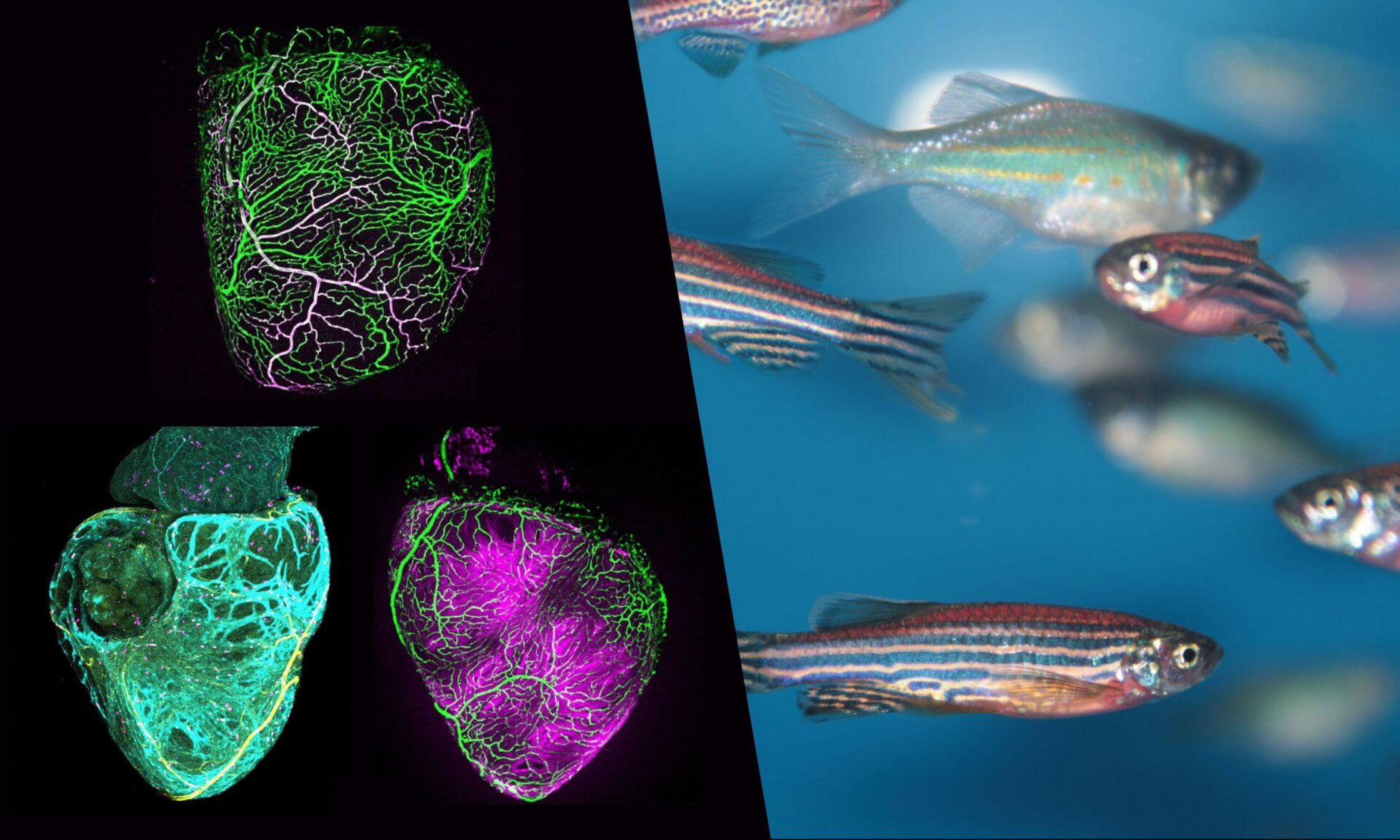
Just in time for Heart Month, the laboratory of University de Montréal medical professor and CHU Sainte-Justine researcher Rubén Marín‑Juez has unveiled the first comprehensive atlas of coronary vessel development in the zebrafish.
Led by PhD student Muhammad Abdul Rouf and the fruit of several years of meticulous analysis, this large‑scale work traces—with unprecedented single‑cell resolution—how vessels form and interact with the cardiac muscle.
Published in Development, the study combines 3D imaging with genetic zebrafish lines to determine where, when and how coronary vessels establish themselves and guide the maturation of cardiac muscle cells.
“For a long time, vessels were viewed simply as conduits that transport blood," Marín‑Juez, a researcher at CHU Sainte‑Justine's Azrieli Research Centre and professor in the university's Faculty of Medicine.
"Today, we show that the vessels orchestrate the growth of the cardiac muscle, starting from the earliest stages of heart formation," he said.
The idea for the research emerged from the lab’s work on cardiac regeneration: in adult zebrafish, coronary vessels form a kind of scaffold that enables the muscle to rebuild after an injury. This observation led the team to trace development back to the embryo to understand how these interactions arise from the very beginning of life.