Stops cells from aging
Telomerase is an enzyme that acts on telomeres, protective caps of repeated DNA sequences at the ends of chromosomes that prevent chromosomes from degrading and preserve genome stability.
Telomeres gradually shorten over time with repeated cell division, and when they become too short, the cell stops dividing— a process known as cellular senescence. This gradual shortening happens in most of the body’s cells and contributes to the natural aging of tissues.
However, during embryonic development and in certain cells, such as germ cells, stem cells and some immune cells, telomere shortening needs be counteracted to slow down senescence.
Enter telomerase: it lengthens telomeres, enabling these cells to undergo extended rounds of division while preserving chromosomal integrity.
In most somatic cells, telomerase is inactive or present only at very low levels. In cancer, however, the enzyme is abnormally reactivated, allowing cells to divide indefinitely—a phenomenon known as cellular immortality. Studies show that telomerase is reactivated in about 90 per cent of cancer cells.
Understanding how telomerase works is therefore essential to understanding the mechanisms behind aging and cancer in humans.
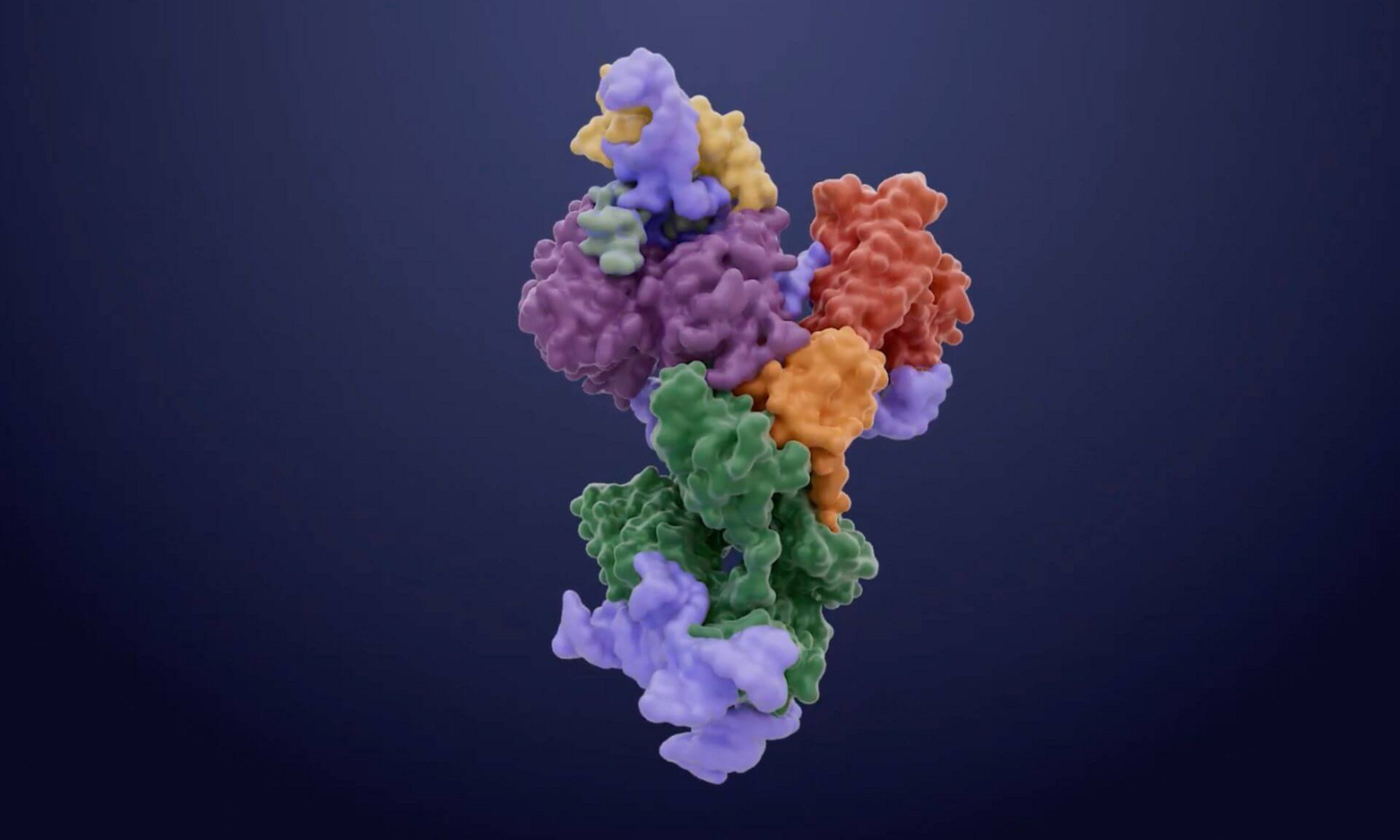
Surprising molecular architecture
In mapping the three-dimensional structure of yeast telomerase, Chartrand and his team made some unexpected discoveries.
For example, they identified what’s known as a zinc finger, a recurring structural pattern often seen in proteins capable of binding to DNA or RNA, but never before observed in telomerase.
“Our research suggests that this zinc finger binds a portion of telomerase’s RNA, thereby stimulating the enzyme’s activity,” explained Chartrand. “To test this hypothesis, we mutated the pattern and telomerase activity disappeared almost completely, confirming the importance of this structure to the enzyme’s functioning.”
The researchers also discovered that a protein called Est3 acts as a molecular scaffold, linking the various components of telomerase together and ensuring structural integrity.
“The Est3 protein is essential for telomerase to remain active in cells,” Chartrand said. This function of Est3 was previously unknown.